Investing in Cell and Gene Therapies (CGT) – Future and Development
Binocs is pleased to announce the appointment of Akshay Peer (Ph.D.). As a co-founder of TrakCel, Akshay has spent his last 8 years working closely with a large number of CGT developers and associated organizations. He was instrumental in growing the company from 2 employees to being an industry-leading player and an established brand in the industry. In the role of director of Binocs business development and sales, Akshay will be part of the Bluecrux US organization and will advise on our strategies for investing in CGT. To begin, he will support the Product and Solutions teams in further developing and enhancing our CGT scheduling capabilities and footprint.
Cell & Gene therapies are the most exciting new methods for targeting previously unmet patient needs including genetic disorders, rare diseases and different types of cancers (blood-related malignancies and solid tumors). They often offer superior treatment options for life-threatening illnesses, and in some cases, have the potential to cure these diseases altogether.
Did you know that Bluecrux has published an eBook about the CGT industry?
Introducing
Voices from the Advanced Therapies Space: collected discussions and advice from experts in the CGT industry
A unique deep dive into the challenges of the cell and gene therapy sector, from the perspective of established experts in the field, in their own words
Download your FREE copy today!The U.S. Food and Drug Administration (FDA) has approved 17 CGT drugs so far, including two advanced cell therapies (CAR-T products) to cure leukemia and lymphoma. The market is further expected to grow and the FDA expects 30-60 approved products by 2030. These transformational therapies will require further scale-up, reduced cost of goods and management to truly make a substantial impact on the masses.
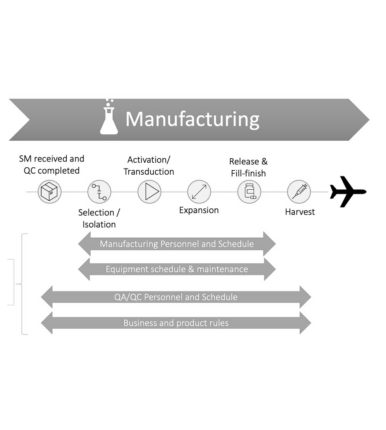
As this tidal wave of new treatments moves closer to making landfall, the CGT industry is still working to overcome the challenges of commercialization such as securing raw materials, finding the right equipment, expertise and all the other issues associated with scaling up production from the lab into the manufacturing plant. The bulk of the current manufacturing for CGT products is being carried out by the Contract Manufacturing Organization (CMO). However, CMO capacity for manufacturing is stretched thin due to high demand. More and more CGT developers are choosing to construct their own facility, several new options have emerged to help drug developers design a space with the right kind of flexibility for the market.
To scale-up production of autologous products efficiently, managing precious resources (technicians, quality, etc) and scheduling manufacturing runs in line with the patient’s starting material collection is critical. Therefore, to support the growth and evolution of this exciting industry, our company is making strategic investments into personnel and technology.
Akshay Peer: “I am very excited to be working with the Binocs team. The scheduling and capacity management solution – Binocs is definitely a requirement for all CGT products as manufacturing capacity is always limited. Patients need to be treated in a very timely manner which includes scheduling all different aspects of their treatment without hassles. Binocs will enable CGT developers to manage capacity at their or contracted manufacturing facilities whilst providing a seamless patient experience at treatment centers. I am looking forward to helping to establish Binocs as a de-facto solution for scheduling and capacity management across the industry.”
Joachim Lasoen, VP of Binocs product: “The past two years we have built a significant Binocs footprint in the area of Small Scale Production and CGT. With our Integrated Production Slot Management solution, we have taken a leadership position in CGT supply chain planning. Bringing Akshay on board accelerates our investment. He will bring a wealth of knowledge and expertise of the operational requirements and challenges in setting and scaling-up CGT product-based supply chains.”
Akshay very well versed with the processes inside and outside the manufacturing facilities, especially for autologous products. He has worked closely with CAR-T, personalized cancer vaccine, Tumour Infiltrating Lymphocyte (TIL), matched-allogeneic and CRISPR Cas-9 based developers. Akshay is also named inventor on three patents for traceability and use of technology in solving difficult business challenges and processes. He obtained a Ph.D. from Swansea University (UK) for applying RFID technology in the construction industry value chain in 2009.
-
Advanced therapy developers face challenges in scaling up their supply chain and manufacturing operations. Learn what the future of CGT holds!Read more
-
Bluecrux publishes its first CGT eBook, putting professionals in the spotlight!
Bluecrux announces the publication of its first CGT eBook, titled “Voices from the Advanced Therapies Space: collected discussions and advice from experts in the CGT industry”Read more -
How to orchestrate the cell and gene therapy supply chain
Orchestrating the cell and gene therapy supply chain is particularly complex. We outline 7 key challenges and solutions provided by Binocs.Read more
Akshay Peer
Akshay joined Bluecrux as the CGT lead in 2021 after a distinguished career as the co-founder of TrakCel (now the market leader in providing CGT orchestration solutions). With more than a decade’s experience in supporting advanced therapy manufacturers involved in CAR-T, TILs, Tregs, CRISPR Cas 9, and other similar ‘made-to-order’ therapies, Akshay has worked closely with the Binocs Product and Solution teams at Bluecrux to engineer a planning solution that is tailored to the specific needs of this challenging industry. Akshay also leads the IWG for Binocs and actively participates in industry events and thought leadership discussions.






