
Digitization, automation and online testing: the future of pharma quality control
Digitization, automation and online testing: The future of pharma quality control
Recently, McKinsey published an interesting article on the vast improvement potential they see in pharma QC labs.
The article positions three possible horizons for which QC leaders can aim:
- Digitally enabled labs
Labs that pull the sample testing in the lab. There is a lot of emphasis on connected instruments, data science, and advanced planning and scheduling. - Automated labs
Labs that have 60-80% of testing in the lab and 20-40% on the shop floor. The focus is on automated testing and non-testing processes. - Distributed quality control
This focuses on real-time testing at the line, enabling review by exception and parametric release.
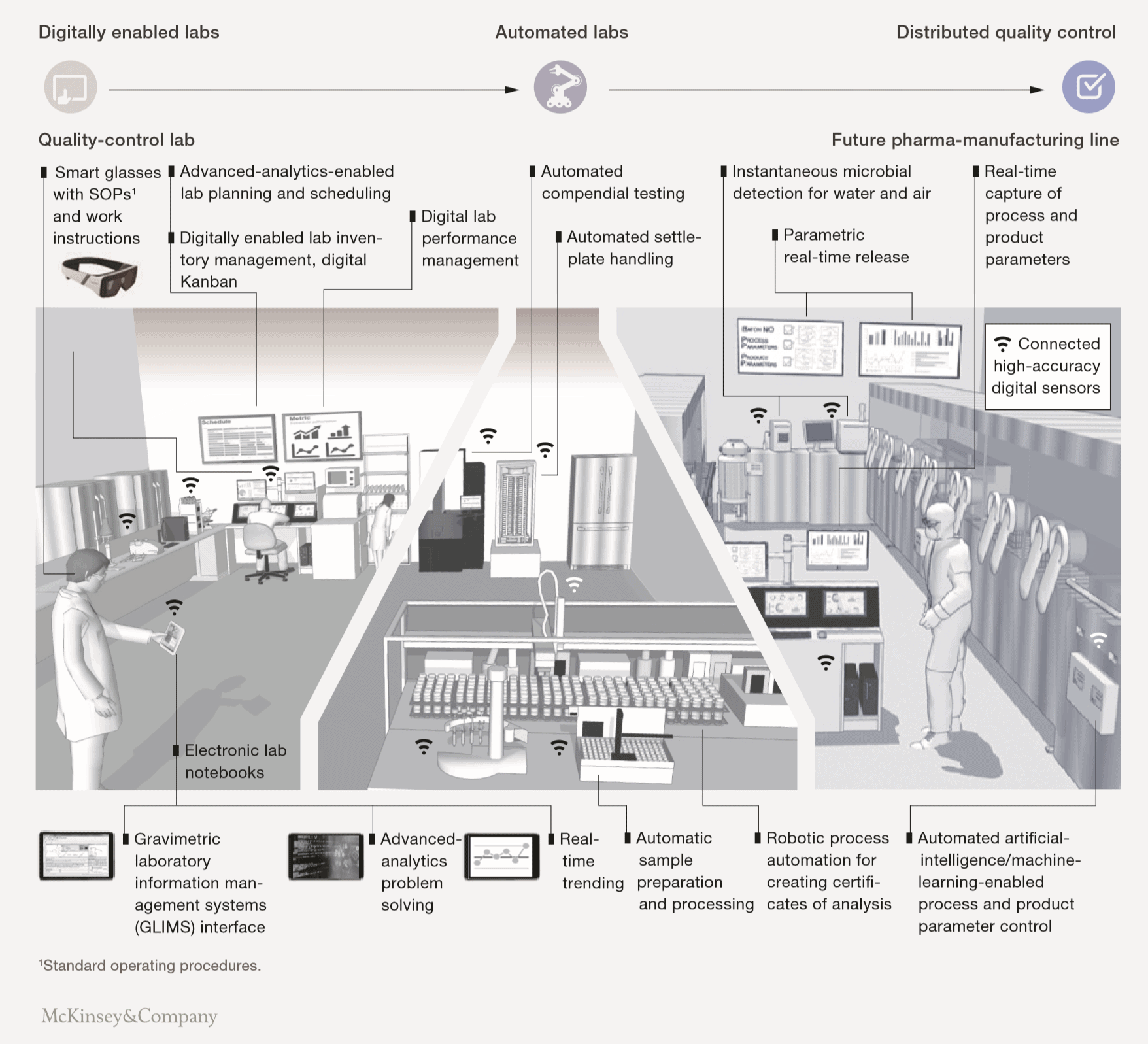
From Binocs we are mainly active in the labs that aim for digital enabling and automation. In this area McKinsey sees important improvement potential:
- Cost reductions of 25% to 40% in digitally enabled labs. This comes from the elimination of documentation work and advanced scheduling
- Reduce lab leadtime with 10% to 20%
- Test Productivity increase of up to 30% by implementing advanced lab scheduling
- Sample taking and preparation productivity increase of up to 80%
- Reduce deviations with 80% through advanced analytics
- Accelerate deviation closing with 90%
How to measure the potential for improvement?
McKinsey sees different root causes for this and based on our experience we couldn’t agree more with their conclusions.
In the past decades, a lot of attention went (and is still going) to manufacturing automation. QC labs have not really been in the spotlights. But as volume volatility and product complexity are increasing, labs don’t cut it anymore with a LIMS and some instrument connections. QC labs become a critical operation in the supply chain and deserve equal attention.
According to McKinsey, QC labs need to develop their vision, business case and transformation roadmap. The QC “pharmacists” need support from “digital engineers” that can help them break through perceived constraints on technological capabilities and system validation requirements.
McKinsey also positions Lab planning and scheduling to accelerate the process. They suggest: “Targeting a fully tested end-to-end future state prototype rather than testing and rapidly scaling up high-value solutions to capture quick wins. For example, schedule automation and optimization can be implemented quickly and start generating significant value even if a lab is not yet mostly paperless and fully digitized“.
And that’s of course where Binocs comes in…
Read the whole article of McKinsey.



